20th March 2026
Published a paper in Nature Communications.
Klara Kohoutova, Dalibor Kosek, Adam Brzezina, Karolina Honzejkova, Veronika Obsilova* & Tomas Obsil*. Structural basis of the cyclin Y/14-3-3 protein-mediated activation of CDK16. Nature Communications (2026). DOI: 10.1038/s41467-026-70778-5. IF = 15.7
* shared corresponding authorship
Link: Nat. Comm. 2026.
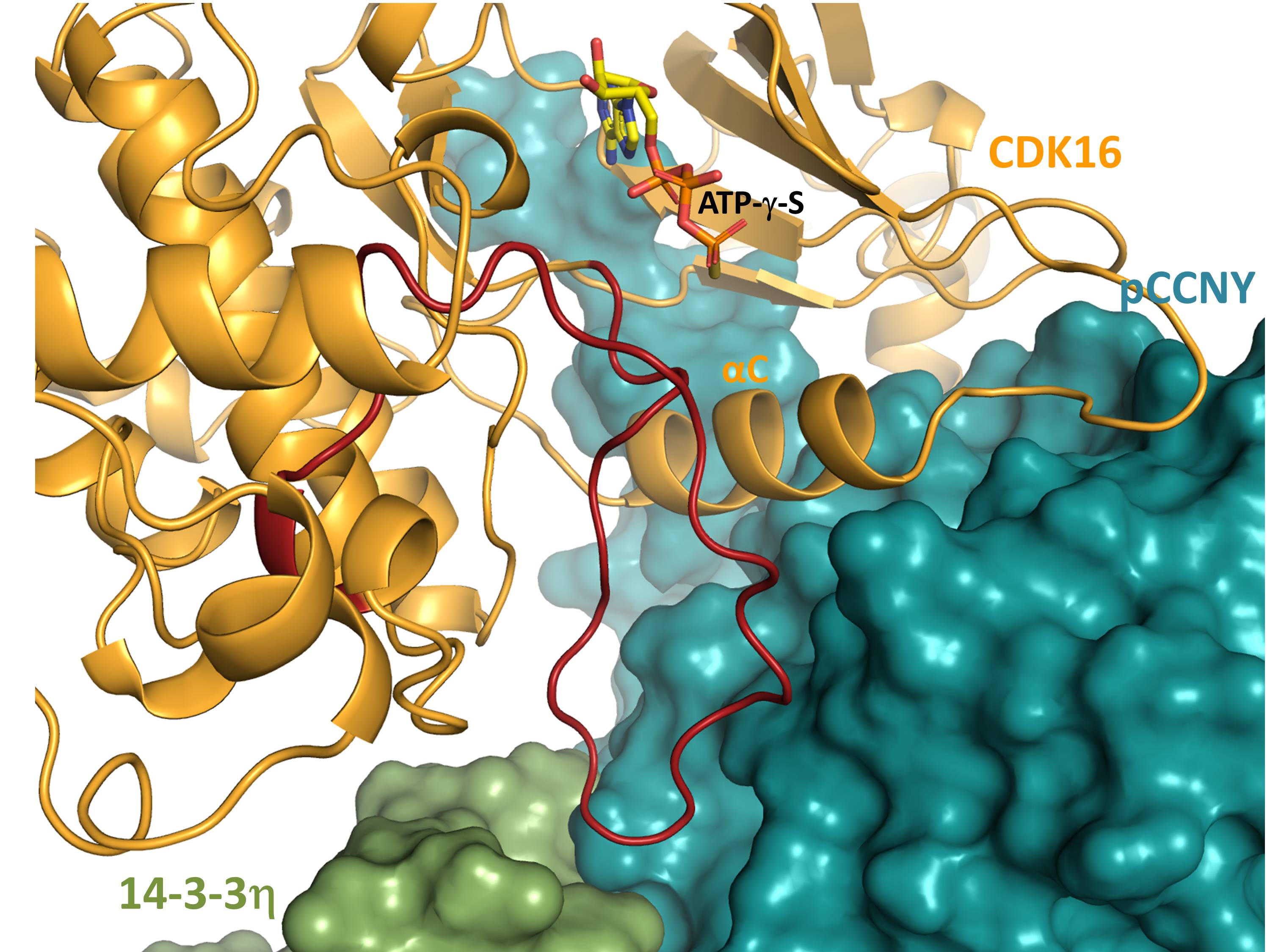
A key region of the binding interface between CDK16, pCCNY and 14-3-3. The activation segment of CDK16 (residues 304-331, shown in dark red) adopts an ordered conformation stabilized by intra- and intermolecular interactions with pCCNY and 14-3-3 (shown in surface representation).
Andrej Tekel received the Czech Society for Structural Biology prize for an outstanding student presentation at the XXII Discussions on Structural Molecular Biology & 9th CIISB User Meeting, held on 19–21 March 2026 in Nové Hrady, for his talk "Structural Mechanisms of Nedd4-2 Autoinhibition and Regulation by Membranes, Calcium and 14-3-3η".
9th January 2026
Published a paper in FEBS Journal.
Masa Janosev, Tomas Obsil & Veronika Obsilova*. Lock, Relax, Load and Shoot: A Molecular Perspective on Nedd4 Regulation. FEBS Journal (2026). DOI: 10.1111/febs.70424. IF = 4.2
Link: FEBS J. 2026
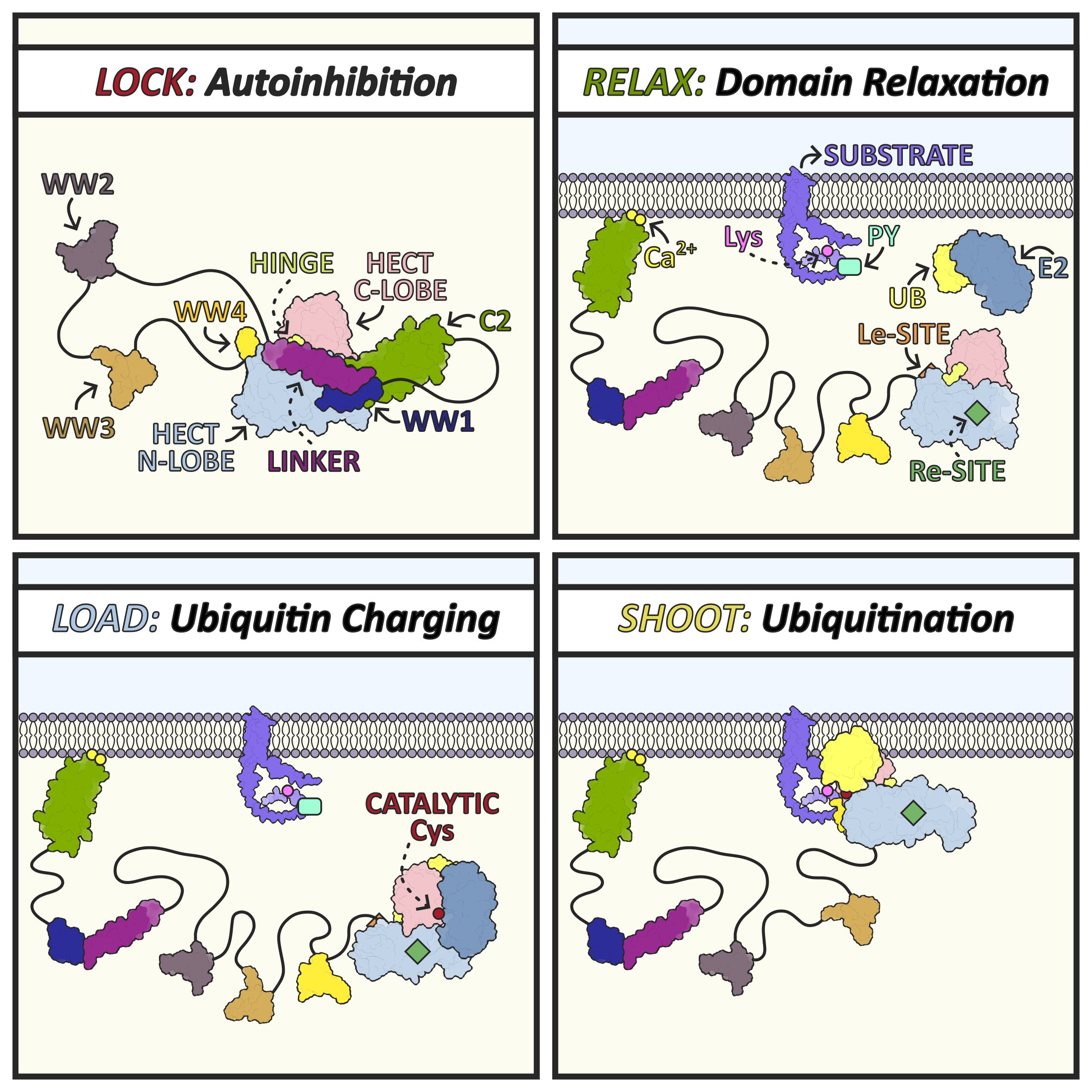
Structural basis of inactive and active states of the Nedd4 HECT E3 ligase subfamily, following a “lock, relax, load, and shoot” mechanism. In the locked, autoinhibited state, intramolecular domain interactions restrain the HECT domain. Relaxation releases these restraints, allowing loading of ubiquitin onto the catalytic cysteine, followed by the “shoot” step of (auto)ubiquitination. Nedd4 ligases regulate diverse cellular pathways, making them important targets for mechanistic and pharmacological studies.
24th June 2025
Klára Kohoutová defended her PhD thesis.
6th June 2025
Martin Hýbl defended his master thesis (M.Sc.).
26th May 2025
Published a paper in Nature Communications.
Masa Janosev, Dalibor Kosek, Andrej Tekel, Rohit Joshi, Karolina Honzejkova, Pavel Pohl, Tomas Obsil* & Veronika Obsilova*. Structural basis of ubiquitin ligase Nedd4-2 autoinhibition and regulation by calcium and 14-3-3 proteins. Nature Communications 16:4875 (2025). DOI: 10.1038/s41467-025-60207-4. IF = 14.7
* shared corresponding authorship
Link: Nat. Comm. 2025, 16, 4875.
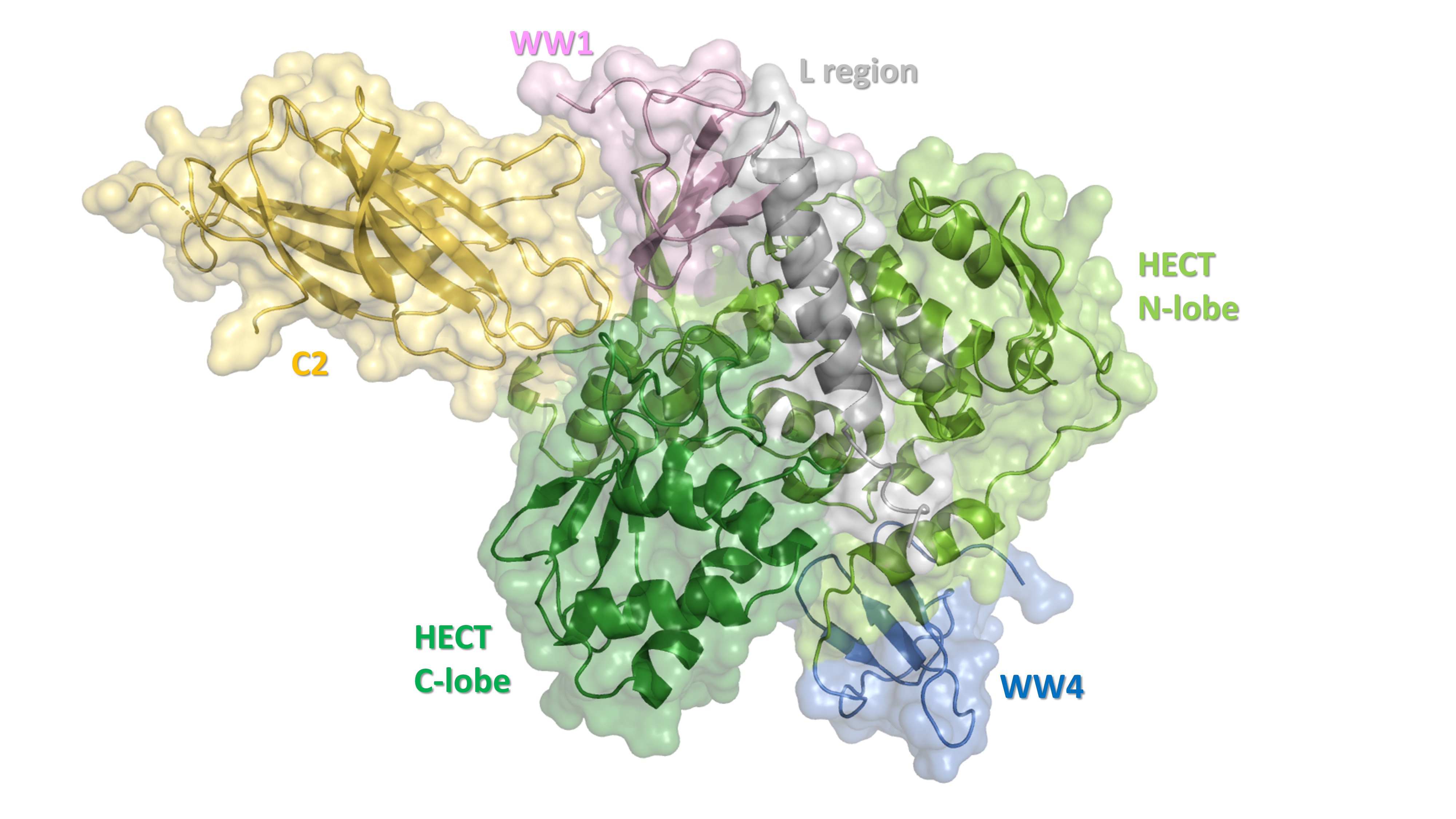
Structure of Nedd4-2 obtained by cryo-EM reconstruction: C2, calcium binding domain (peach); WW1 (pale purple); L region (gray); WW4 (blue); HECT N-lobe, N-lobe of the catalytic HECT domain (light green); HECT C-lobe, C-lobe of the catalytic HECT domain (dark green).
27th May 2025
Published a paper in Nature Communications.
Klara Kohoutova, Pavel Srb, Veronika Obsilova*, Vaclav Veverka* & Tomas Obsil*. Structural plasticity of the FOXO-DBD:p53-TAD interaction. Nature Communications 16:4907 (2025). DOI: 10.1038/s41467-025-59106-5. IF = 14.7
* shared corresponding authorship
Link: Nat. Comm. 2025, 16, 4907

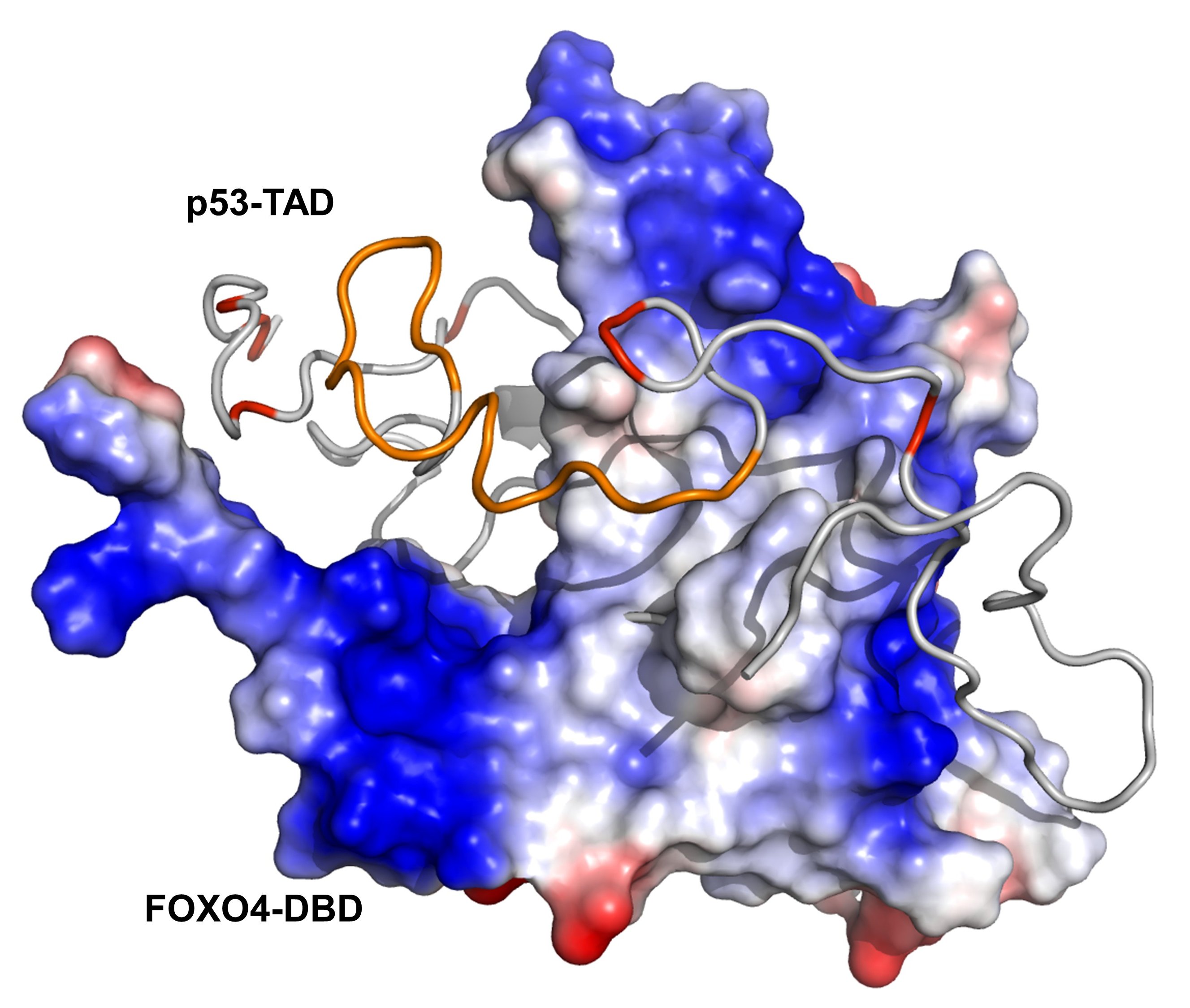

Structure of one of the conformers of the FOXO4-DBD:p53-TAD complex. The surface of FOXO4-DBD is colored according to the electrostatic potential. The p53-TAD protein is shown as a grey ribbon, the TAD2 region is highlighted in orange. Negatively charged Glu and Asp residues of p53-TAD are highlighted in red
27th March 2024
Published a paper in eLife.
Karolina Honzejkova, Dalibor Kosek, Veronika Obsilova* and Tomas Obsil*. The cryo-EM structure of ASK1 reveals an asymmetric architecture allosterically modulated by TRX1. eLife 13:RP95199 (2024). DOI: 10.7554/eLife.95199. IF = 6.4
* shared corresponding authorship
Link: eLife 2024, 13, RP95199
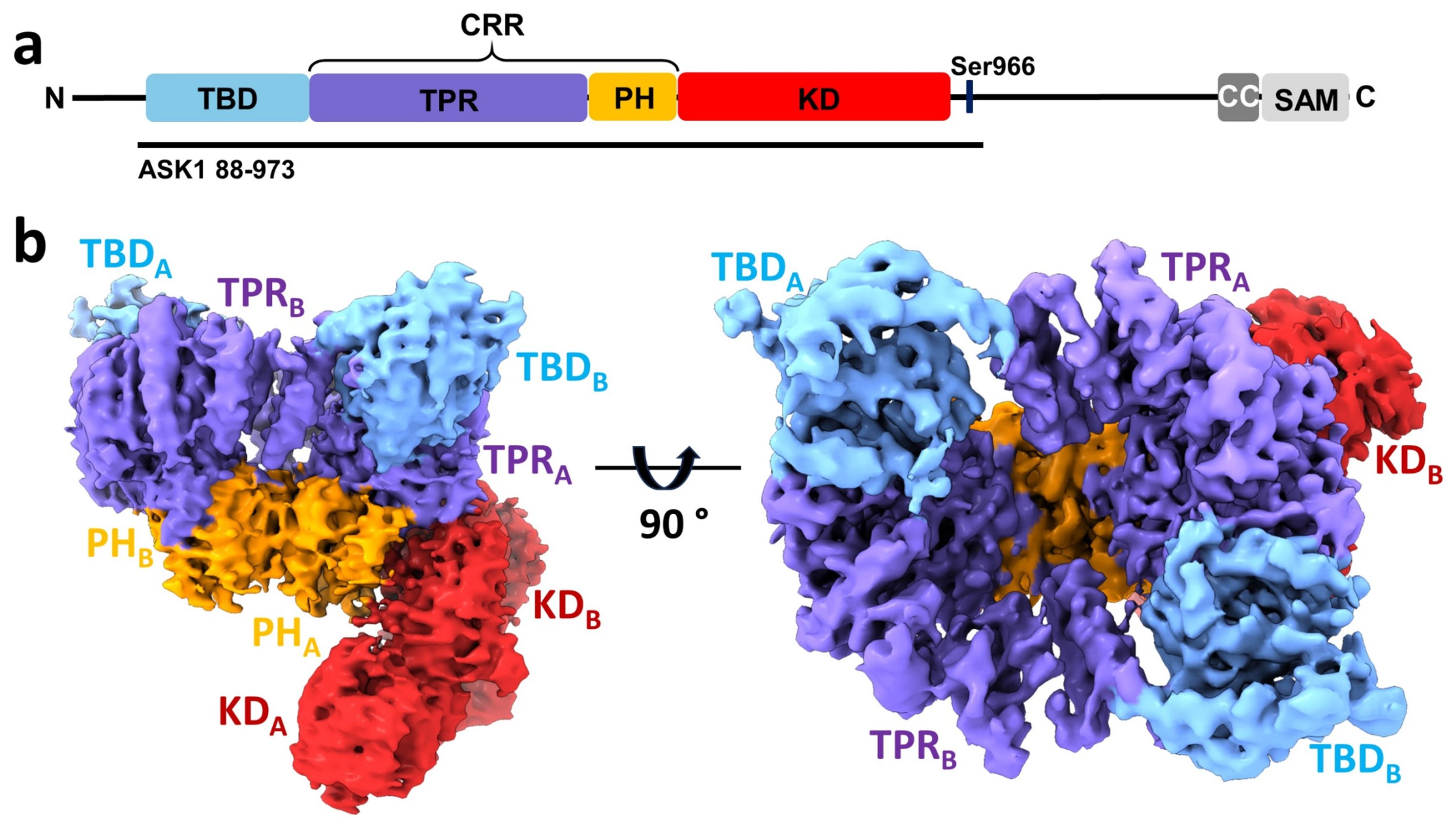
(a) Domain structure of ASK1. TBD, thioredoxin-binding domain; TPR, tetratricopeptide repeat domain; PH, pleckstrin-homology domain; CRR, central regulatory region; KD, kinase domain; CC, coiled-coil motif; SAM, sterile alpha motif domain. (b) Cryo-EM density map of ASK1.